Bone remains from 5,500 years ago provide new clues about the evolution of a global pathogen

![]() 14ymedio, Federico Kukso/SINC Agency, January 24, 2026 — Some 5,500 years ago, a hunter died about 30 kilometers southwest of what is now Bogotá, Colombia. He wasn’t tall—barely five feet three inches—and was a little over 45 years old, a considerable age for his time. Following the rites of his people, his family placed his body in a rock shelter that, centuries later, would be called Tequendama I.
14ymedio, Federico Kukso/SINC Agency, January 24, 2026 — Some 5,500 years ago, a hunter died about 30 kilometers southwest of what is now Bogotá, Colombia. He wasn’t tall—barely five feet three inches—and was a little over 45 years old, a considerable age for his time. Following the rites of his people, his family placed his body in a rock shelter that, centuries later, would be called Tequendama I.
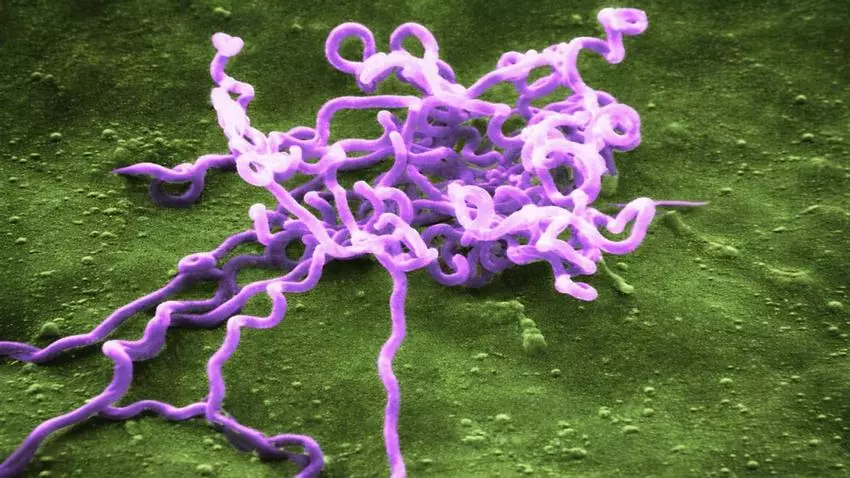
No one knows this man’s name, what his voice sounded like, or the exact cause of his death. The only thing scientists have been able to confirm, after meticulous paleogenetic analysis, is that his skeleton held a secret all that time. “To our surprise, we found DNA from the bacterium Treponema pallidum, responsible for several diseases such as syphilis, yaws, and bejel, in a leg bone,” Colombian archaeologist Miguel Delgado, one of the authors of the research, told SINC.
In a study published in the journal Science, this researcher from the National University of La Plata (Argentina) and a team of Italian, American, German, and Swiss geneticists reveal that this is the oldest known molecular evidence of this pathogen in humans on a global scale. The finding redefines what was previously known about one of the infectious diseases that has most affected populations worldwide.
“Understanding the evolutionary history of this disease helps us to understand it in depth,” adds Delgado, known for his study of the peopling of the Americas. “It allows us to better understand its manifestation and its current spread.”
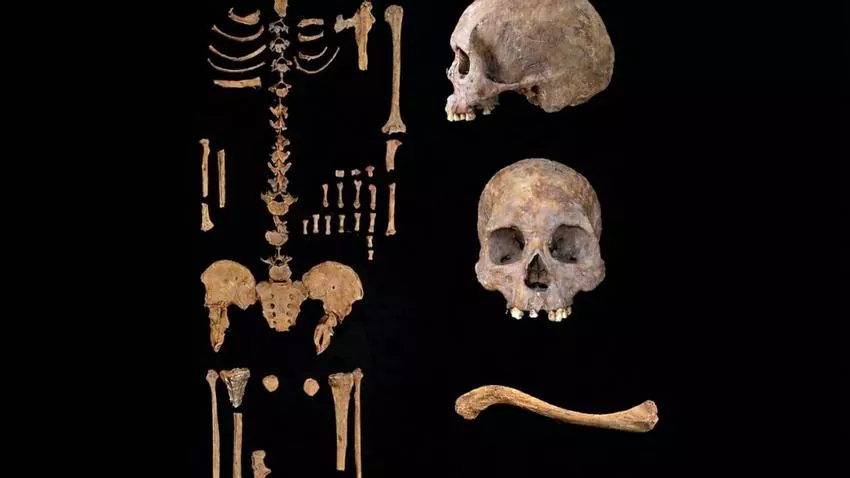
The Tequendama I archaeological site is located in the Bogotá Savannah in the eastern highlands of Colombia, a region that played a key role in the early human expansion in South America. Since 1969, archaeologists like Gonzalo Correal Urrego have recovered stone tools, rock art, animal bones, and hundreds of well-preserved human remains there, some dating back as much as 12,000 years. Among them is the bodyof this 1.58-meter-tall man, identified as TE1-3.
Radiocarbon dating indicates that he lived approximately 5,500 years ago. There are no clear signs as to the cause of his death: no evidence of violence or complex diseases. “It was probably a natural death, a consequence of an extremely harsh lifestyle and a poor diet,” the archaeologist suspects.
The discovery of this bacterium’s DNA was, in a way, fortuitous, scientists explain. To reconstruct the evolutionary history of populations and the diseases they suffered, paleogeneticists usually examine DNA preserved in the inner ear or teeth, where the greatest amount of prehistoric genetic material is preserved. “However, when we took the samples, we had neither teeth nor a skull,” notes Delgado, a scientist at CONICET in Argentina.
After obtaining a license from the Colombian Institute of Anthropology and History (ICANH) – the regulatory body for archaeological heritage in Colombia – the researchers decided to take the risk and sent samples of this hunter’s tibia to different laboratories to perform ancient DNA analysis.
First, they sent the sample to the Human Paleogenomics Laboratory of German biological anthropologist Lars Fehren-Schmitz at the University of California, USA. Then, they sent it to the Population Genomics Group of paleogeneticist Anna-Sapfo Malaspinas at the University of Lausanne, Switzerland. Upon sequencing the material, the scientists noticed a type of DNA that did not belong to the individual, but rather to a helical bacterium: Treponema pallidum.
“It was truly surprising,” says molecular anthropologist Elizabeth A. Nelson of Southern Methodist University in Dallas, one of the authors of the article. “Recovering DNA from ancient pathogens is always a challenge, and Treponema pallidum is particularly difficult to detect. This is because, during the stages of skeletal lesion formation, the pathogen is usually present in the body at very low levels.”
This discovery extends the complex evolutionary history of this pathogen: it is three thousand years older than the human remains with T. pallidum found on the southeast coast of Brazil – reported in 2024 – and five thousand years earlier than the arrival of Europeans to the continent.
It is also one of the sexually transmitted infections that causes the most concern.
The geographic origin, evolution, and spread of treponemal diseases—each caused by subspecies of the diverse Treponema family of microbes —remain one of the most persistent debates in the history of infectious diseases. Without treatment, syphilis can lead to devastating neurological consequences: blindness, deafness, mental disorders, and even death.
It is also one of the sexually transmitted infections that causes the most concern: according to the World Health Organization, its global incidence is increasing year after year. Yaws, on the other hand, primarily affects the skin and bones and is transmitted through direct contact in warm, humid regions. Bejel and pinta, meanwhile, cause mouth ulcers and bone lesions.
Where and when the pathogens that have affected human societies first appeared remains a great mystery. Recent technological advances and reduced sequencing costs have enabled unprecedented study of the evolution of bacteria, viruses, and parasites.
In particular, research into ancient DNA (aDNA) of pathogens provides unique information about past microbial infections. This genetic material acts as a time capsule: it possesses remarkable stability, can be analyzed even after millennia, and allows us to observe how pathogens have genetically changed over time.
“The field of ancient DNA took off with the arrival of high-throughput sequencing in 2008,” recalls Argentine molecular biologist Nicolás Rascován, from the Pasteur Institute in Paris. “In 2011, the genome of an ancient pathogen was recovered for the first time from archaeological samples: that of the bacterium Yersinia pestis, responsible for the plague. It was then that the study of past diseases through DNA sequencing gained momentum.”
“By studying the DNA of ancient pathogens, we can trace their evolution as they developed.”
From that moment on, the burgeoning science of paleogenomics made it possible to reconstruct the deep history of the pathogens responsible for human infectious diseases, such as tuberculosis (Mycobacterium tuberculosis), smallpox (Variola virus), and hepatitis B (HBV). Various studies show that these infections emerged around 6,500 years ago, coinciding with the beginning of close contact between humans and domesticated animals, a change that increased the risk of zoonosis, that is, the transmission of pathogens from animals to people.
“By studying the DNA of ancient pathogens, we can trace their evolution as they developed,” explains Italian computational biologist Davide Bozzi of the University of Lausanne. “This can give us insights into how pathogens adapted to infect humans or how they became virulent. This knowledge is an essential first step in understanding and controlling the emergence of new viruses and bacteria or the reemergence of past diseases.”
In the spring of 1495, the French king Charles VIII invaded the Kingdom of Naples, driven by his desire to become emperor. After a brief period of success, his mercenary troops began to succumb to an invisible enemy: a disfiguring disease previously unknown to the world. Medical chronicles of the time describe fever, widespread skin eruptions, genital ulcers, intense bone pain, and a high mortality rate.
The French called it “the Neapolitan disease.” For the Italians, it was “morbus gallicus” (French disease). The Russians knew it as “the Polish disease,” and the Poles, “the German disease.” For the inhabitants of Flanders and North Africa, it was “the Spanish disease,” while for the Spanish, it was “the buboes.”
Within a few months, the disease spread across Europe along military and trade routes, leaving many survivors with neurological damage and permanent deformities. Today, this episode is considered the first documented historical record of syphilis in the Old World.
Some believed it was divine punishment. For astrologers, the epidemic was a consequence of two solar eclipses and the conjunction of Mars and Saturn. Others, however, blamed the sailors who returned from America with Christopher Columbus in 1493. “It is possible that 15th-century syphilis was the first globalized emerging infectious disease and a harbinger of all subsequent ones, from HIV/AIDS to COVID-19,” say anthropologists Molly K. Zuckerman and Lydia Bailey of Mississippi State University.
The term syphilis was coined only in 1530 by the Italian physician and poet Girolamo Fracastoro, in his poem Syphilis sive Morbus Gallicus (Syphilis, or the French Disease). In it, the shepherd Syphilius offends the god Apollo and, as punishment, unleashes a plague that ravages his people. The disease is named after him.
“It is possible that syphilis in the 15th century was the first globalized emerging infectious disease and a harbinger of all subsequent ones, from HIV/AIDS to COVID-19.”
Since then, several competing hypotheses have sought to explain its origin. The “Columbian hypothesis” maintains that syphilis was already widespread in the human populations of the Americas before the European conquest. The “pre-Columbian hypothesis,” on the other hand, states that the bacterium T. pallidum circulated in Eurasia during the medieval period and possibly earlier, but was mistaken for other diseases. The current consensus is that it was present on both continents, but in the Americas, a variant evolved into venereal syphilis. European contact would have accelerated its rapid global spread.
“The question of the origin of disease has been raised for centuries because people wanted to blame someone,” notes anthropologist Nasreen Broomandkhoshbacht of the University of California. “These narratives are harmful; they can be used to justify violence and the dehumanization of entire groups of people.”
Since its discovery in 1905, this bacterium has intrigued microbiologists, especially because it cannot be cultured in the laboratory. In 1943, penicillin revolutionized the treatment of syphilis. In 1998, its entire genome was sequenced. At the time, it was thought that this achievement would pave the way for the development of preventative vaccines. That has not happened.
Ancient T. pallidum DNA, identified in the tibia of this hunter-gatherer who lived 5,500 years ago in Colombia, provides valuable clues to understanding the evolution of the disease. By comparing this genetic material with that of modern strains of the same pathogen, it is possible to trace the changes it has undergone over time. In this case, phylogenetic analysis reveals that the genome of this bacterium corresponds to a previously unknown branch of Treponema pallidum, which diverged before the common ancestor of the current subspecies.
Although it clearly falls within the T. pallidum species, the TE1-3 strain exhibits remarkable genetic diversity and is distinct from modern strains. “What we found cannot be classified as syphilis,” cautions Broomandkhoshbacht.
“Syphilis, bejel, and yaws are very different diseases, and all are much more closely related to each other than to the pathogen we described in our research. We now know that in the past there was an even greater diversity of these pathogens, which could mean there was even greater variation in their behavior. Historical records of syphilis show that it has changed dramatically over the last 500 years, so even if what we found was a direct ancestor of syphilis (which it isn’t), we could assume that it behaved quite differently,” he adds.
The findings also suggest that T. pallidum predates the emergence of agriculture in the Americas. “This individual lived about 5,500 years ago, in a Middle Holocene hunter-gatherer community, long before the intensification of agriculture or the demographic changes of the colonial era—two factors often associated with the spread of infectious diseases,” Nelson adds.
In contrast, the TE1-3 lineage appears to be linked to the social and ecological conditions typical of hunter-gatherer societies: high mobility, interactions in small communities, and close contact with wild animals.
Even so, scientists find it striking that the remains of the studied individual show no visible signs of disease. “We cannot determine whether the absence of skeletal pathology reflects an early or latent phase of the infection,” they state.
Many questions remain unanswered. “We still don’t know how this ancient lineage was transmitted, whether animals played a role as reservoirs in the past, or whether the pathogen we identified represents an early or extinct lineage,” explains Elizabeth Nelson.
In addition to continuing their research in Colombia, the scientific team is expanding the scope of their work southward into the Andes, in collaboration with Peruvian colleagues. “To understand how the Treponema lineage identified in Tequendama evolved, it is essential to study other individuals over time and in different regions,” says anthropologist Lars Fehren-Schmitz. “Likewise, to better understand the ecology of diseases, research should not be limited to the remains of human ancestors, but should also extend to the analysis of animal remains.”
____________
COLLABORATE WITH OUR WORK: The 14ymedio team is committed to practicing serious journalism that reflects Cuba’s reality in all its depth. Thank you for joining us on this long journey. We invite you to continue supporting us by becoming a member of 14ymedio now. Together we can continue